|
|
Introduction to Corrosion Monitoring |
View PDF version 
|
What is Corrosion Monitoring?
The field of corrosion measurement, control, and prevention covers a very broad spectrum of technical activities. Within the sphere of corrosion control and prevention, there are technical options such as cathodic and anodic protection, materials selection, chemical dosing and the application of internal and external coatings. Corrosion measurement employs a variety of techniques to determine how corrosive the environment is and at what rate metal loss is being experienced. Corrosion measurement is the quantitative method by which the effectiveness of corrosion control and prevention techniques can be evaluated and provides the feedback to enable corrosion control and prevention methods to be optimized.
A wide variety of corrosion measurement techniques exists, including:
| Non Destructive Testing Analytical Chemistry |
|
Analytical Chemistry |
|
• Ultrasonic testing
• Radiography
• Thermography
• Eddy current/magnetic flux
• Intelligent pigs |
• pH measurement
• Dissolved gas (O2, CO2, H2S)
• Metal ion count (Fe2+, Fe3+)
• Microbiological analysis |
|
|
|
|
|
|
|
| Operational Data |
Fluid Electrochemistry |
|
• pH
• Flow rate (velocity)
• Pressure
• Temperature |
• Potential measurement
• Potentiostatic measurements
• Potentiodynamic measurements
• A.C. impedance |
|
|
|
|
|
|
| Corrosion Monitoring |
|
|
• Weight loss coupons
• Electrical resistance
• Linear polarization
• Hydrogen penetration
• Galvanic current |
|
|
|
|
|
|
|
|
|
Some corrosion measurement techniques can be used on-line, constantly exposed to the process stream, while others provide off-line measurement, such as that determined in a laboratory analysis. Some techniques give a direct measure of metal loss or corrosion rate, while others are used to infer that a corrosive environment may exist.
Corrosion monitoring is the practice of measuring the corrosivity of process stream conditions by the use of "probes" which are inserted into the process stream and which are continuously exposed to the process stream condition.
Corrosion monitoring "probes" can be mechanical, electrical, or electrochemical devices.
Corrosion monitoring techniques alone provide direct and online measurement of metal loss/corrosion rate in industrial process systems.
Typically, a corrosion measurement, inspection and maintenance program used in any industrial facility will incorporate the measurement elements provided by the four combinations of on-line/offline, direct/indirect measurements.
- Corrosion Monitoring Direct, On-line
- Non Destructive Testing Direct, Off-line
- Analytical Chemistry Indirect, Off-line
- Operational Data Indirect, On-line
In a well controlled and coordinated program, data from each source will be used to draw meaningful conclusions about the operational corrosion rates with the process system and how these are most effectively minimized.
|
| |
The Need for Corrosion Monitoring
The rate of corrosion dictates how long any process plant can be usefully and safely operated. The measurement of corrosion and the action to remedy high corrosion rates permits the most cost effective plant operation to be achieved while reducing the life-cycle costs associated with the operation.
Corrosion monitoring techniques can help in several ways:
- by providing an early warning that damaging process conditions exist which may result in a corrosion-induced failure.
- by studying the correlation of changes in process parameters and their effect on system corrosivity.
- by diagnosing a particular corrosion problem, identifying its cause and the rate controlling parameters, such as pressure, temperature, pH, flow rate, etc.
- by evaluating the effectiveness of a corrosion control/prevention technique such as chemical inhibition and the determination of optimal applications.
- by providing management information relating to the maintenance requirements and ongoing condition of plant.
|
| |
Corrosion Monitoring Techniques
A large number of corrosion monitoring techniques exist. The following list details the most common techniques which are used in industrial applications:
- Corrosion Coupons (weight loss measurements)
- Electrical Resistance (ER)
- Linear Polarization Resistance (LPR)
- Galvanic (ZRA) / Potential
- Hydrogen Penetration
- Microbial
- Sand/Erosion
Other techniques do exist, but almost all require some expert operation, or otherwise are not sufficiently rugged or adaptable to plant applications.
Of the techniques listed above, corrosion coupons, ER, and LPR form the core of industrial corrosion monitoring systems. The four other techniques are normally found in specialized applications which are discussed later.
These corrosion monitoring techniques have been successfully applied and are used in an increasing range of applications because:
- The techniques are easy to understand and implement.
- Equipment reliability has been demonstrated in the field environment over many years of operational application.
- Results are easy to interpret.
- Measuring equipment can be made intrinsically safe for hazardous area operation.
- Users have experienced significant economic benefit through reduced plant down time and plant life extension.
|
| |
Corrosion Coupons (Weight Loss)
The Weight Loss technique is the best known and simplest of all corrosion monitoring techniques. The method involves exposing a specimen of material (the coupon) to a process environment for a given duration, then removing the specimen for analysis. The basic measurement which is determined from corrosion coupons is weight loss; the weight loss taking place over the period of exposure being expressed as corrosion rate.
The simplicity of the measurement offered by the corrosion coupon is such that the coupon technique forms the baseline method of measurement in many corrosion monitoring programs.
The technique is extremely versatile, since weight loss coupons can be fabricated from any commercially available alloy. Also, using appropriate geometric designs, a wide variety of corrosion phenomena may be studied which includes, but is not limited to:
- Stress-assisted corrosion
- Bimetallic (galvanic) attack
- Differential aeration
- Heat-affected zones
Advantages of weight loss coupons are that:
- The technique is applicable to all environments - gases, liquids, solids/particulate flow.
- Visual inspection can be undertaken.
- Corrosion deposits can be observed and analyzed.
- Weight loss can be readily determined and corrosion rate easily calculated.
- Localized corrosion can be identified and measured.
- Inhibitor performance can be easily assessed.
In a typical monitoring program, coupons are exposed for a 90-day duration before being removed for a laboratory analysis. This gives basic corrosion rate measurements at a frequency of four times per year. The weight loss resulting from any single coupon exposure yields the "average" value of corrosion occurring during that exposure. The disadvantage of the coupon technique is that, if a corrosion upset occurs during the period of exposure, the coupon alone will not be able to identify the time of occurrence of the upset, and depending upon the peak value of the upset and its duration, may not even register a statistically significant increased weight loss.
Therefore, coupon monitoring is most useful in environments where corrosion rates do not significantly change over long time periods. However, they can provide a useful correlation with other techniques such as ER and LPR measurements.
|
| |
Electrical Reistance (ER) Monitoring
ER probes can be thought of as "electronic" corrosion coupons. Like coupons, ER probes provide a basic measurement of metal loss, but unlike coupons, the value of metal loss can be measured at any time, as frequently as required, while the probe is in-situ and permanently exposed to the process stream.
|
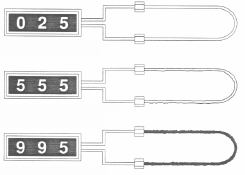 |
The wire loop represented here is new with its total diameter of 40 mils (total useful probe life of 10 mils).
Here the element has experienced about 5 mils penetration or about half of its useful life. The increased electrical resistance of the element will register as 5 mils metal penetration into the piping or process system.
Here the element has measured 10 mils of penetration and requires replacement. |
|
| |
| The ER technique measures the change in Ohmic resistance of a corroding metal element exposed to the process stream. The action of corrosion on the surface of the element produces a decrease in its cross-sectional area with a corresponding increase in its electrical resistance. The increase in resistance can be related directly to metal loss and the metal loss as a function of time is by definition the corrosion rate. Although still a time averaged technique, the response time for ER monitoring is far shorter than that for weight loss coupons. The graph below shows typical response times. |
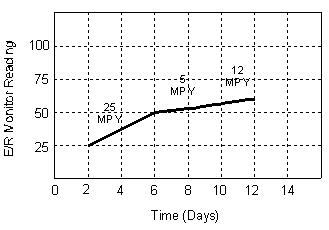
|
ER probes have all the advantages of coupons, plus:
- They are applicable to all working environments gases, liquids, solids, particulate flows.
- Direct corrosion rates can be obtained.
- Probe remains installed in-line until operational life has been exhausted.
- They respond quickly to corrosion upsets and can be used to trigger an alarm.
ER probes are available in a variety of element geometries, metallurgies and sensitivities and can be configured for flush mounting such that pigging operations can take place without the necessity to remove probes. The range of sensitivities allows the operator to select the most dynamic response consistent with process requirements.
|
| |
Linear Polarization Resistance (LPR) Monitoring
The LPR technique is based on complex electro-chemical theory. For purposes of industrial measurement applications it is simplified to a very basic concept. In fundamental terms, a small voltage (or polarization potential) is applied to an electrode in solution. The current needed to maintain a specific voltage shift (typically 10 mV) is directly related to the corrosion on the surface of the electrode in the solution. By measuring the current, a corrosion rate can be derived.
The advantage of the LPR technique is that the measurement of corrosion rate is made instantaneously. This is a more powerful tool than either coupons or ER where the fundamental measurement is metal loss and where some period of exposure is required to determine corrosion rate. The disadvantage to the LPR technique is that it can only be successfully performed in relatively clean aqueous electrolytic environments. LPR will not work in gases or water/oil emulsions where fouling of the electrodes will prevent measurements being made.
|
| |
Galvanic/Potential Monitoring
- The galvanic monitoring technique, also known as Zero Resistance Ammetry (ZRA) is another electrochemical measuring technique. With ZRA probes, two electrodes of dissimilar metals are exposed to the process fluid. When immersed in solution, a natural voltage (potential) difference exits between the electrodes. The current generated due to this potential difference relates to the rate of corrosion which is occurring on the more active of the electrode couple.
Galvanic/Potential monitoring is applicable to the following electrode couples:
- Bimetallic corrosion
- Crevice and pitting attack
- Corrosion assisted cracking
- Corrosion by highly oxidizing species
- Weld decay
Galvanic current measurement has found its widest applications in water injection systems where dissolved oxygen concentrations are a primary concern. Oxygen leaking into such systems greatly increases galvanic currents and thus the corrosion rate of steel process components. Galvanic monitoring systems are used to provide an indication that oxygen may be invading injection waters through leaking gaskets or deaeration systems.
|
| |
Specialized Monitoring
Biological Monitoring
Biological monitoring and analysis generally seeks to identify the presence of Sulphate Reducing Bacteria - SRB's. This is a class of anaerobic bacteria which consume sulphate from the process stream and generate sulphuric acid, a corrosive which attacks production plant materials.
Sand / Erosion Monitoring
These are devices which are designed to measure erosion in a flowing system. They find wide application in oil/gas production systems where particulate matter is present.
Hydrogen Penetration Monitoring
In acidic process environments, hydrogen is a by-product of the corrosion reaction. Hydrogen generated in such a reaction can be absorbed by steel particularly when traces of sulphide or cyanide are present. This may lead to hydrogen induced failure by one or more of several mechanisms. The concept of hydrogen probes is to detect the amount of hydrogen permeating through the steel by mechanical or electrochemical measurement and to use this as a qualitative indication of corrosion rate. |
| |
Instrumentation
There exists a variety of instrument options associated with the various corrosion monitoring techniques. Three classifications are:
- Portable
- Continuous Single Channel
- Continuous Multi-Channel
In some applications such as those experienced in oil/gas production and refining, instrumentation is required to be certified for use in "hazardous areas". For portable instruments this is most often achieved by having the equipment certified as "intrinsically safe" by a recognized authority such as BASEEFA (U.K.), U.L. (U.S.A.), or CENELEC (Europe). For hardwired continuous monitoring electronics, isolation barriers can be used to ensure that, in the event of a fault condition, insufficient energy is transmitted to the hazardous field area so that an explosive spark cannot be produced.
|
| |
Probe Fitting Styles
There are two fundamental fitting styles for corrosion probes: fixed and removable under pressure.
Fixed styles of probes/sensors have typically a threaded or flanged attachment to the process plant. For fixed styles of sensors, removal can only be accomplished during system shut down or by isolation and depressurization of the sensor location.
From time to time, corrosion coupons and probes require removal and replacement. It is sometimes more convenient to be able to remove and install sensors while the process system is operational. To facilitate this, there are two distinct systems which permit removal/installation under pressure.
In refinery and process plant environments where pressures are normally less than 1500 psi, a Retractable System is used. This consists of a packing gland (stuffing box) and valve arrangement. For environments such as those experienced in oil/gas production where pressures of several thousand psi are experienced, a special High Pressure Access System is used. This permits the safe and easy installation/removal of corrosion monitoring devices at working pressures up to 3600 psi.
|
| |
Applications of Corrosion Monitoring Techniques
Corrosion monitoring is typically used in the following situations:
- Where risks are high - high pressure, high temperature, flammable, explosive, toxic processes.
- Where process upsets can cause high corrosivity.
- Where changes in operating conditions can cause significant changes in corrosion rate.
- Where corrosion inhibitors are in use.
- In batch processes, where corrosive constituents are concentrated due to repeated cycling.
- Where process feedstock is changed.
- Where plant output or operating parameters are changed from design specifications.
- In the evaluation of corrosion behavior of various alloys.
- Where induced potential shifts are used to protect systems and/or structures.
- Where product contamination due to corrosion is a vital concern.
Corrosion monitoring may be used in virtually any industry where corrosion prevention is a primary requirement. Some examples of industries and specific areas of interest include, but are not limited to:
Oil/Gas Production
- Flowlines
- Gathering Systems
- Transport Pipelines
- Water Injection Facilities
- Vessels
- Processing
- Water Systems
- Chemical Injection Systems
- Drilling Mud Systems
- Water Wash Systems
- Desalters
|
|
Refining
- Crude Overheads
- Visbreakers
- Vacuum Towers
- Sour Water Strippers
- Amine Systems
- Cooling Systems
|
Pulp and Paper
- Digesters
- White Liquor
- Boiler Systems
|
|
Utilities
- Cooling Systems
- Effluent Systems
- Make-Up Water Systems
- Boiler Water Systems
|
Petrochemicals/Chemicals/Processing
- Process Systems
- Cooling Systems
|
|
|
In any corrosion monitoring system, it is common to find two or more of the techniques combined to provide a wide base for data gathering. The exact techniques which can be used depend on the actual process fluid, alloy system, and operating parameters.
Corrosion monitoring offers an answer to the question of whether more corrosion is occurring today as compared to yesterday. Using this information it is possible to qualify the cause of corrosion and quantify its effect. Corrosion monitoring remains a valuable weapon in the fight against corrosion, thereby providing substantial economic benefit to the user. |
| |
|
|
|

